Why
does ice float?
Ice floats because
it is less dense than water. Water has a density of 1.0 gm/cubic cm. The
density of ice Ih is 0.931 gm/cubic cm. But,
why is ice less dense than water if both are made up of molecules of H2O? | |
|
| [Ice
structure shown using the JMol Applet] | Try
this!! Click the right mouse button with the cursor over the image--> Render
--> Schemes --> CPK Spacefill Click on the left mouse button
and rotate the ice structure. Do you see the open spaces in ice? They are
formed at low temperatures when water molecules form many stable hydrogen
bonds. Compare this structure to that below of water at room
temperature. | | |
| [Water
Molecules are shown using the JMol Applet] | Render
--> Schemes --> CPK Spacefill Hydrogen bond lengths between
water molecules will vary since the molecules are in constant motion, unlike ice
which is a rigid lattice structure (above) To measure distance Double
click on one atom then drag to second atom-- double click again
To
measure angles Double click on first atom, once on middle atom, twice
on atom three. |
| 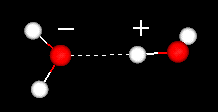
| Liquid
water has a partially ordered structure in which hydrogen
bonds are constantly being formed and breaking up. | The
strong hydrogen bonds give water a high cohesiveness and, consequently, surface
tension. This is evident when small quantities of water are put onto a nonsoluble
surface and the water stays together as drops.
On the other
hand ice has a rigid lattice structure. In liquid water each molecule is
hydrogen bonded to approximately 3.4 other water molecules. In ice each
each molecule is hydrogen bonded to 4 other molecules. Compare the two
structures below. Notice the empty spaces within the ice structure. In
ice Ih, each water forms four hydrogen bonds with O---O distances of 2.76 Angstroms
to the nearest oxygen neighbor. Because of ordered structure in ice there are
less H20 molecules in a given space of volume. Try this --
1) Measure the O-O distances between any two adjacent oxygen atoms
in ice shown in the above structure.. Please
enter your answer in the space provided:
2) Measure the O-O-O angle formed between adjacent oxygen
atoms in ice. 3) What is the length of the hydrogen bond
H-O in ice?
| 
