What are Atomic Orbitals?
HISTORICAL PERSPECTIVES OF THE ATOM After J.J. Thomson's discovered the electron in 1897, it became clear that atoms were not the smallest building blocks of nature, but rather were composite particles. The newly discovered structure within atoms tempted many to imagine what the atoms structure might look like. Thomson theorized that multiple electrons revolved in orbit-like rings within a positively charged jelly-like substance, The newly discovered structure within atoms tempted many to imagine how the atom's constituent parts might interact with each other. Thomson theorized that multiple electrons revolved in orbit-like rings within a positively charged jelly-like substance. This theory known as the "plum pudding model" was the most widely accepted explanation of atomic structure until 1909. In 1904, the Japanese physicist Omura Nagaoka developed an early planetary model -- The Saturnian Model of the atom--. Nagaoka's model was based around an analogy to the explanation of the stability of the Saturn rings (the rings are stable because the planet they orbit is very massive). The model made two predictions: a very massive nucleus (in analogy to a very massive planet) electrons revolving around the nucleus, bound by electrostatic forces (in analogy to the rings revolving around Saturn, bound by gravitational forces). Both predictions were successfully confirmed by Rutherford (who mentions Nagaoka's model in his 1911 paper in which the nucleus is proposed).Nagaoka himself however recognized a fundamental defect in the theory even at its conception, namely that a classical charged object cannot sustain orbital motion because it is accelerating and therefore loses energy due to electromagnetic radiation
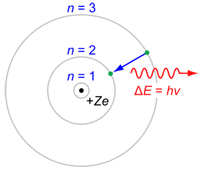
With de Broglie's suggested the existence of electron matter waves in 1924, and Heisenberg's discovery of his uncertainty relation,Max Born suggested that the electron's position needed to be described by a probability distribution which was connected with finding the electron at some point in the wave-function which described its associated wave packet. The new quantum mechanics did not give exact results, but only the probabilities for the occurrence of a variety of possible such results. Heisenberg held that the path of a moving particle has no meaning if we cannot observe it, as we cannot with electrons in an atom. In the quantum picture of Heisenberg, Schrödinger and others, the Bohr atom number n for each orbital became known as ann-sphere in a three dimensional atom and was pictured as the mean energy of the probability cloud of the electron's wave packet which surrounded the atom. WHAT ARE ATOMIC ORBITALS?
VISUALIZING ATOMIC ORBITALS The atomic orbitals of the hydrogen atom can be visualized as a cloud around the nucleus. The orbital represents a probability of finding the electron at a particular location. Regions that have a higher concentration of dots signify a greater probablility. Atomic orbitals do not always have the shape of a sphere. Higher orbitals have very unusual shapes.
The pdb coordinates used for the Jmol files were retrieved from: 3-D Graphic Examples of Atomic Orbitals, where you can find additional examples. |
||||
Related Activies
- Atoms - Elements - Compounds
- Mass Volume Density
- Molecular Modeling-- An NGSS Activity
- Water and Ice Module
- States of Matter
- How do molecules of solids, liquids and gas behave differently?
- Science of Fluids
- Buoyancy and Archimedes Principle
- Speed Velocity and Acceleration
- Gifted and Talented STEM
- EDinformatics Science Challenge
- How does a battery work?