Chemical Bonds
A single water molecule is made up of two atoms of hydrogen and one atom of oxygen.
The atoms are joined by chemical bonds which are forces of attraction that hold atoms together. It is the bonds between atoms that give molecules different properties than the atoms they are composed of.
Covalent Bonds
A covalent bond is a bond that is formed when two atoms share electrons
The Lewis Dot Structure is often used to represent sharing of electrons in bonds.
A single bond is formed when 1 pair of electrons is shared; a double bond is formed when 2 pairs of electrons are shared; and a triple bond is formed when 3 pairs of electrons are shared.
Ionic Bonds
Table salt is made up of two very elements that can be very dangerous, namely, sodium and chlorine. Combined they form a safe compound.
Sodium and chlorine combine during a reaction that will transfer one valence electron from sodium to chlorine.
After the transfer sodium and chlorine are no longer neutral. The sodium and chlorine have becomes ions (atoms with unequal number of protons and electrons). The chlorine now has a net charge of negative 1 (-1) and the sodium ion has a charge of +1. Since objects of opposite charge attract each other, the two ions will be held tightly together. This type of attraction is called an ionic bond.
In a crystal of sodium chloride, each ion will have six neighboring ions of opposite charge. This makes ionic bonding a very strong type of interaction between atoms. These strong bonds ionic compounds a high melting point.
Hydrogen Bonds
Hydrogen bonds are formed when a hydrogen atom is shared between two molecules
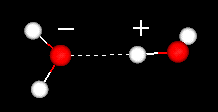
Hydrogen bonds have polarity. The hydrogen atom above (+) in water is covalently attached to a very electronegative oxygen atom (to right). It also shares its partial positive charge with a second electronegative oxygen atom (left).
Related Activies
- Mass Volume Density
- Molecular Modeling-- An NGSS Activity
- Water and Ice Module
- States of Matter
- How do molecules of solids, liquids and gas behave differently?
- Science of Fluids
- Buoyancy and Archimedes Principle
- Speed Velocity and Acceleration
- Gifted and Talented STEM
- EDinformatics Science Challenge
- How does a battery work?
 Click for 3-D image
Click for 3-D image