Surface Tension and Capillary Action
WHAT IS SURFACE TENSION?
Surface tension is a property of liquids that arises from unbalanced molecular cohesive forces at or near a surface. At an air water interface the surface tension results from the greater attraction of water molecules to each other (due to cohesion) than to the molecules in the air (due to adhesion). The net effect is an inward force at its surface that causes water to behave as if its surface were covered with a stretched elastic membrane. Because of the relatively high attraction of water molecules for each other, water has a high surface tension.
Surface tension arises from the strong interactions between water molecules, called hydrogen bonding. It is this strong interaction which also manifests in the other unusual property of water such as its high boiling point.
Surface tension of water also manifests as the so-called hydrophobic effect. Hydrophobic molecules tend to be non-polar and, thus, prefer other neutral molecules and non-polar solvents--"water- hating". A hydrophilic molecule or portion of a molecule is one that has a tendency to interact with or be dissolved by water and other polar substances --"water-loving" -- See also: How does soap work?
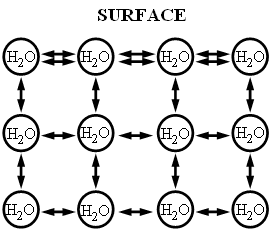 |
The cohesive forces between molecules in a liquid are shared with all neighboring molecules. Those on the surface have no neighboring molecules above and, thus, exhibit stronger attractive forces upon their nearest neighbors on and below the surface. For molecules in center of water, all attractive forces are balanced. On the surface, molecules have unbalanced surface ... fluid tries to minimize it's surface area. This is why water forms a drop. the surf race tries to minimize area and inside liquid is under pressure. image edited from USGS.gov |
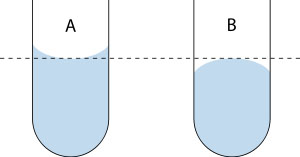 |
The meniscus is the curve in the upper surface of a liquid close to the surface of the container or another object. It is caused by surface tension. It can be either convex or concave, depending on the liquid and the surface. A shown on Left -- a concave meniscus occurs when the particles of the liquid are more strongly attracted to the container than to each other, causing the liquid to climb the walls of the container. This occurs between water and glass. B shown on Right -- a convex meniscus occurs when the particles in the liquid have a stronger attraction to each other than to the material of the container. Convex menisci occur, for example, between mercury and glass in barometers. Note: Cohesive attraction or cohesive force is the action or property of similar molecules sticking together, being mutually attractive. Cohesion, along with adhesion (attraction between unlike molecules), helps explain phenomena such as meniscus, surface tension and capillary action. image edited from Reading the Meniscus (Jleedev) Wikipedia. |
Examples of surface tension in action include the following:
--formation of liquid droplets,
--the ability of a needle to float on water,
--why bubbles are round
--soap being used the break up water tension.
Surface tension has the dimension of force per unit length, or of energy per unit area. The two are equivalent—but when referring to energy per unit of area, people use the term surface energy—which is a more general term in the sense that it applies also to solids and not just liquids.
Surface tension, usually represented by the symbol γ, is measured in force per unit length. Its SI unit is newton per meter.
In terms of energy: surface tension --gamma-- of a liquid is the ratio of the change in the energy of the liquid to the change in the surface area of the liquid (that led to the change in energy).
![]()
for a derivation of the formula see: wikipedia surface tension
This work W can be interpreted as being stored as potential energy. Thus, surface tension can be also measured in the SI system as joules per square meter. Since mechanical systems try to find a state of minimum potential energy, a free droplet of liquid water will try to assume a spherical shape, which has the minimum surface area for a given volume.
WHAT IS CAPILLARY ACTION?
| TRY THIS: FILL A GLASS WITH DILUTED GRAPE JUICE AS SHOWN BELOW. FOLD A PAPER TOWEL TO CONNECT THE TWO GLASSES...OBSERVE WHAT HAPPENS OVER SEVERAL HOURS... | ||
 |
 |
|
AFTER 20 MINUTES -- WATER IS MOVING UP THE PAPER TOWEL |
AFTER 2 HOURS -- MOSTLY WATER HAS MOVED INTO THE SECOND GLASS |
AFTER 4 HOURS -- SOME PIGMENT HAS MOVING |
Capillary action occurs because water molecules bond each other strongly due to forces of cohesion and adhesion where water molecules are attracted and stick to other substances such as glass or paper. Adhesion of water to the surface of a material will cause an upward force on the liquid. The surface tension acts to hold the surface intact. Capillary action occurs when the adhesion to the surface material is stronger than the cohesive forces between the water molecules. The height to which capillary action will take water is limited by surface tension and gravity.
Notice in the photos above the effect that gravity has on capillary action. Water being a polar molecule and low mass moves easily up the paper. The diluted grape juice contains several non-polar red pigments which are not very soluble in water and do not migrate with the solvent.
If one takes a small capillary tube an inserts it in water and the tube does not have a vacuum like a barometer but is open at top, water will start to rise up. Water wants to stick to the glass and surface tension will push the water up, until the force of gravity prevents further rise.
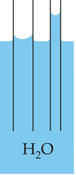 |
Capillarity is the result of cohesion of water molecules and adhesion of those molecules to a solid material. In the case of a glass tube inserted in water with openings at both ends, as the edges of the tube are brought closer together, such as in a very narrow tube, the liquid will be drawn upward in the tube. The more narrow the tube, the greater the rise of the liquid. Greater surface tension and increased ratio of adhesion to cohesion also result in greater rise. |
Since for water in a tube all values are constant except r, radius, the height of rise is solely dependent on the radius of the tube. |
Capillary action is due to the pressure of cohesion and adhesion which cause the liquid to work against gravity.
Capillary action (sometimes capillarity, capillary motion, or wicking) is the ability of a liquid to flow in narrow spaces without the assistance of, and in opposition to, external forces like gravity.CAPILLARY ACTION IN PLANTS
Capillary action is the process that plants use to pull water and mineral up from the ground. It is is the movement of liquid along a surface of a solid caused by the attraction of molecules of the liquid to the molecules of the solid. The molecules of the water (liquid) are attracted to the molecules inside the stem similar to capillary action of water in a glass tube.
There are three forces involved with the process of capillary action in plants.
1) Adhesion, the process of attracting two dissimilar molecules. For plants, adhesion allows for the water to stick to the organic tissues of plants.
2)Cohesion keeps similar molecules together. For plants, cohesion keeps the water molecules together.
3)Surface tension is a property of liquids that arises from unbalanced molecular cohesive forces at or near a surface..
TEST YOUR UNDERSTANDING
Mass Volume Density Activities
Mass: Learn how to measure the mass of an object using a triple beam balance
Mass vs. Weight: Mass and weight are often confused by many students. Learn the difference and try some challenging problems.
Volume: Measure volume using a graduated cylinder.
Density of a Solid: Learn to calculate the density of an unknown solid from knowing its mass and volume.
Density of a Liquid: Learn to calculate the density of an unknown liquid from knowing its mass and volume using a graduated cylinder and triple beam balance. Learn what a hydrometer is, and what it can do.
Density Challenge: Great page for gifted and talented students! Some excellent challenging problems.
Assessment: Twenty questions on mass, volume and density (two levels of difficulty). Your test is marked online.
Science Project Ideas: Ideas for science projects using mass, volume and density concepts learned from this module.
Mass Volume Density Lab Exercise: Problem: What is the relationship between water pressure and depth of water?
An Integrated Math Science and Art (STEAM) Activity- Mass, Volume Density Activity using the Gates Project from Central Park NYC.

