Compounds and Molecules
A compound is a pure substance composed of two or more different atoms chemically bonded to one another. A compound can be destroyed by chemical means. It might be broken down into simpler compounds, into its elements or a combination of the two, whereas the same techniques do not cause an >element to break down.
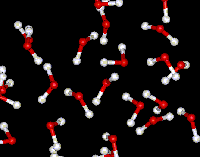 |
Microscopic view of the molecules of the compound water (liquid phase). Oxygen atoms are red and hydrogen atoms are white. |
A compound consists of atoms of two or more different elements bound together, can be broken down into a simpler type of matter (elements) by chemical means (but not by physical means), has properties that are different from its component elements, and always contains the same ratio of its component atoms.
A molecule consists of two or more atoms of the same element, or different elements, that are chemically bound together. A molecule is the smallest particle in a chemical element or compound that has the chemical properties of that element or compound. Molecules are made up of atoms that are held together by chemical bonds.
EXAMPLES OF IONIC AND COVALENT COMPOUNDS:
|
Ionic Compound-- Ionic compounds are defined as being compounds where two or more ions are held next to each other by electrical attraction. The formation of an Ionic bond is the result of the transfer of one or more electrons from a metal onto a non-metal. The result is that one of the ions will have a positive charge (called a "cation") and the other will have a negative charge ("anion"). Cations are usually metal atoms and anions are either nonmetals or polyatomic ions (ions with more than one atom). All ionic compounds form crystals. Ionic compounds have a definite melting point and contain ionic bonds.When dissolved in water they will conduct electricity. An example would be sodium chloride (NaCl). To view the 3-D structure of a salt crystal click here. |
|
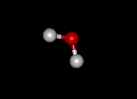
Covalent Compound-- A covalent compound is a compound in which the atoms that are bonded share electrons rather than transfer electrons from one to the other. While ionic compounds are usually formed when metals bond to nonmetals, covalent compounds are formed when two nonmetals bond to each other. The water molecule shown to the left is composed of two different types of atoms covalently bound: oxygen (red) and hydrogen (white). To view the 3-D structure of water click here. |
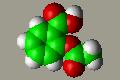 |
Covalent Compound-- The aspirin molecule shown to the left is composed of three different types of atoms: carbon (green), oxygen (red) and hydrogen (white) covalently bound. To view the 3-D structure of aspirin click here. |
Related Activies
- Mass Volume Density
- Molecular Modeling-- An NGSS Activity
- Water and Ice Module
- States of Matter
- How do molecules of solids, liquids and gas behave differently?
- Science of Fluids
- Buoyancy and Archimedes Principle
- Speed Velocity and Acceleration
- Gifted and Talented STEM
- EDinformatics Science Challenge
- How does a battery work?