Hydrogen bonds...
See also
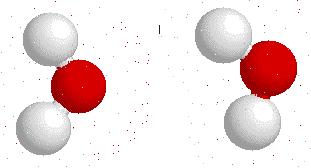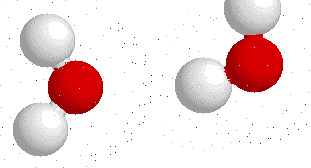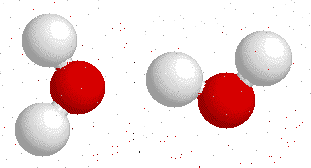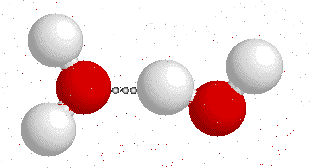
Hydrogen bonds --->>The water dimer
In chemistry, a hydrogen bond is a type of attractive intermolecular force that exists between two partial electric charges of opposite polarity. Although stronger than most other intermolecular forces, the hydrogen bond is much weaker than both the ionic bond and the covalent bond. Within macromolecules such as proteins and nucleic acids, it can exist between two parts of the same molecule, and figures as an important constraint on such molecules' overall shape.
As the name "hydrogen bond" implies, one part of the bond involves a hydrogen atom. The hydrogen must be attached to a strongly electronegative heteroatom, such as oxygen, nitrogen or fluorine, which is called the hydrogen-bond donor. This electronegative element attracts the electron cloud from around the hydrogen nucleus and, by decentralizing the cloud, leaves the atom with a positive partial charge. Because of the small size of hydrogen relative to other atoms and molecules, the resulting charge, though only partial, nevertheless represents a large charge density. A hydrogen bond results when this strong positive charge density attracts a lone pair of electrons on another heteroatom, which becomes the hydrogen-bond acceptor.
The hydrogen bond is not like a simple attraction between point charges, however. It possesses some degree of orientational preference, and can be shown to have some of the characteristics of a covalent bond. This covalency tends to be more extreme when acceptors bind hydrogens from more electronegative donors.
Strong covalency in a hydrogen bond raises the questions: "To which molecule or atom does the hydrogen nucleusbelong?" and "Which should be labelled 'donor' and which 'acceptor'? According to chemical convention, the donor generally is that atom to which, on separation of donor and acceptor, the retention of the hydrogen nucleus (or proton) would cause no increase in the atom's positive charge. The acceptor meanwhile is the atom or molecule that would become more positive by retaining the positively charged proton. Liquids that display hydrogen bonding are called associated liquids.
Hydrogen bond in water
The most ubiquitous, and perhaps simplest, example of a hydrogen bond is found between water molecules. In a discrete water molecule, water has two hydrogen atoms and one oxygen atom. Two molecules of water can form a hydrogen bond between them. The oxygen of one water molecule has two lone pairs of electrons, each of which can form a hydrogen bond with hydrogens on two other water molecules. This can repeat so that every water molecule is H-bonded with four other molecules (two through its two lone pairs, and two through its two hydrogen atoms.)
- H-O-H...O-H2
Liquid water's high boiling point is due to the high number of hydrogen bonds each molecule can have relative to its low molecular mass. Water is unique because its oxygen atom has two lone pairs and two hydrogen atoms, meaning that the total number of bonds of a water molecule is four. (For example, hydrogen bromide- which has two lone pairs on the Br atom but only one H atom - can have a total of only two bonds.)
- H-Br...H-Br...H-Br
In ice, the crystalline lattice is dominated by a regular array of hydrogen bonds which space the water molecules farther apart than they are in liquid water. This accounts for water's decrease in density upon freezing. In other words, the presence of hydrogen bonds enables ice to float, because this spacing causes ice to be less dense than liquid water.
Were the bond strengths more equivalent, one might instead find the atoms of two interacting water molecules partitioned into two polyatomic ions of opposite charge, specifically hydroxide and hydronium.(Hydronium ions are also known as 'hydroxonium' ions).
- H-O- H3O+
Indeed, in pure water under conditions of standard temperature and pressure, this latter formulation is applicable only rarely; on average about one in every 107 molecules gives up a proton to another water molecule, in accordance with the value of the dissociation constant for water under such conditions.
Hydrogen bond in proteins
Hydrogen bonding also plays an important role in determining the three-dimensional structures adopted by proteins and nucleic acids. In these macromolecules, bonding between parts of the same macromolecule cause it to fold into a specific shape, which helps determine the molecule's physiological or biochemical role. The double helical structure of DNA, for example, is due largely to hydrogen bonding between the base pairs, which link one complementary strand to the other and enable replication.
In proteins, hydrogen bonds form between the backbone oxygens and amide hydrogens. When the spacing of the amino acid residues participating in a hydrogen bond occurs regularly between positions i and i+4, an alpha helix is formed. When the spacing is less, between positions i and i+3, then a 310 helix is formed. When two strands are joined by hydrogen bonds involving alternating residues on each participating strand, a beta sheet is formed.(See also protein folding).
Dihydrogen bond
In a very recent development, hydrogen bonds have been noted between two hydrogen atoms having opposite polarity. An example occurs in the molecule H3NBH3 where the hydrogen atoms on nitrogen have a partial positive charge and the hydrogen atoms on boron have a partial negative charge. The resulting BH...HN attractions cause the molecule to be a solid material rather than a gas as is the case in the closely related substance, H3CCH3. Because two hydrogen atoms are involved, this is termed a dihydrogen bond. See references below (Crabtree, et al.).
Symmetric hydrogen bond
Symmetric hydrogen bonds have been observed recently spectroscopically in formic acid at high pressure (>GPa). Each hydrogen atom forms a partial covalent bond with two atoms rather than one. Symmetric hydrogen bonds have been postulated in ice at high pressure (ice-X). See references below (Goncharov, et al.)
The hydrogen bond remains a fairly mysterious object in the theoretical study of quantum chemistry and physics. Most generally, the hydrogen bond can be viewed as a metric dependent electrostatic scalar field between two or more intermolecular bonds. This is slightly different than the intramolecular bound states of, for example, covalent or ionic bonds; however, hydrogen bonding is generally still a bound state phenomenon, since the interaction energy has a net negative sum. The question of the relationship between the covalent bond and the hydrogen bond remains largely unsettled, though the initial theory proposed by Linus Pauling suggests that the hydrogen bond has a partial covalent nature. While alot of experimental data has been recovered for hydrogen bonds in water, for example, that provide good resolution on the scale of intermolecular distances and molecular thermodynamics, the kinetic and dynamical properties of the hydrogen bond in dynamical systems remains largely mysterious.
References
- A New Intermolecular Interaction: Unconventional Hydrogen Bonds with Element-Hydride Bonds as Proton Acceptor Robert H. Crabtree, Per E. M. Siegbahn, Odile Eisenstein, Arnold L. Rheingold, and Thomas F. Koetzle Acc. Chem. Res. 1996, 29(7), 348 - 354.
- Polymerization of Formic Acid under High Pressure Alexander F. Goncharov, M. Riad Manaa, Joseph M. Zaug, Richard H. Gee, Laurence E. Fried, and Wren B. Montgomery Phys. Rev. Lett. 2005, 94, 065505.
Electricity, Magnetism and Electromagnetism
- Electricity and Magnetism Home
- Electric Charge
- Coulombs Law and Inverse Square Law
- Static Electricity
- The Flow of Electricity
- Electric Current
- Electric Circuits
- Electric Power
- The Nature of Magnets
- Earth as a Magnet
- Magnetism in Action
- Magnetism from Electricity
- Electricity from Magnetism
- How do Transformers Work?
- Ohms Law Lab
- Magnetic Forces Lab
- Why Metals Conduct Electricity
- How does a Battery Work
- How does an Electric Motor Work
Science of Fluids
- Science of Fluids
- What are Fluids?
- What is Pressure?
- What is Hydrostatic Pressure?
- Surface Tension and Capillary Action
- Pascals Principle
- Archimedes Principle
- What is Viscosity?
- Bernouili's Principle