|
The
following activities target a middle school population
although activities are approachable by more advanced
elementary school students. The activities are also ideal
for high school students to review basic concepts.
All
exercises make use of javascript's which provides for
a more interactive environment.
|
|
|
Water can
exist as a solid (ice), liquid (water liquid), or gas (water
vapor). The basic molecular formula for the water molecule is
the same in each H2O. But, as the temperature of the system
changes the hydrogen bonds between water molecules change drastically.
What
is a hydrogen bond?
In chemistry,
a hydrogen
bond is a type of attractive intermolecular force that exists
between two partial electric charges of opposite polarity. Although
stronger than most other intermolecular forces, the hydrogen
bond is much weaker than both the ionic
bond and the covalent bond.
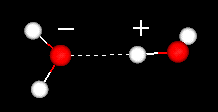 |
Two
water molecules showing their hydrogen bond. For more details
see: the
water dimer. |
What
are the properties of a solid?
A substance in a solid phase is relatively rigid, has a definite
volume and shape.
The atoms
or molecules that comprise a solid are packed close together
and are not compressible.
Because
all solids have some thermal energy, its atoms do vibrate. However,
this movement is very small and very rapid, and cannot be observed
under ordinary conditions. Read
more about states of matter.
|
|
Hydrogen
bonds in ice -- The hydrogen bonds in ice ....
|
What
are the properties of a Liquid?
Liquids
have a definite volume, but are able to change their shape by
flowing.
Liquids
are similar to solids in that the particles touch. However the
particles are able to move around.
Since
particles are able to touch the densities of liquid will be
close to that of a solid.
Since
the liquid molecules can move they will take the shape of their
container. Read
more about states of matter.
What
are the specific properties of liquids?
|
|
Hydrogen
bonds in liquid water
To show hydrogen bonds in water molecules:
Right Click on mouse button:
Select ---> All
Style --> Hydrogen Bonds --> Calculate
Style --> Hydrogen Bonds --> On
|
What
are the properties of a Gas?
Gases have
no definite volume or shape. If unconstrained gases will spread
out indefinitely. If confined they will take the shape of their
container. This is because gas particle have enough energy to
overcome attractive forces. Each of the particles are well separated
resulting in a very low density.
We can calculate an approximate value for number of water molecules
in the above box when in a gas state using the following information:
Avogadros number 6 x 1023
saturation density of water at 20 o C = 17.8 g/m3(
source)
1 mole of water = 18 g
1 cu m= 10-30 cu. nm.
A water molecule is considered to be in the vapor phase if
its distance from the center of particle exceeded 6 nm (60 Angstroms)..
http://pubs.acs.org/doi/abs/10.1021/jp076442f
Read
more about states of matter.
|
|
Gas
molecules are too far apart to form hydrogen bonds.
To calculate the distance between the two molecules shown in the gas phase, double click on one -- drag mouse to other molecule then double click again.
|
|

