| WATER
DIMER
Why does this happen?
Why do the oxygen atoms continue
to be attracted to hydrogen atoms? Remember water is a polar molecule....
A water molecule is formed when two
atoms of hydrogen bond covalently with an atom of oxygen. In a
covalent bond electrons are shared between atoms. In water the
sharing is not equal. The oxygen atom attracts the electrons more
strongly than the hydrogen.This gives water an asymmetrical distribution
of charge. Molecules that have ends with partial negative and
positive charges are known as polar molecules. It is this polar
property that allows water to separate polar solute molecules
and explains why water can dissolve so many substances.
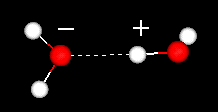
The positive regions in one water
will attract the negatively charged regions in other waters. The
dashes show the hydrogen bond. In a hydrogen bond a hydrogen atom
is shared by two other atoms.
Return to the Water Module
|