| Part
I -Geometry Part
II-Mathematical Relationships
Part
III- Overview of MathMol Tutorials Numerous
connections are found between mathematics and molecular modeling. Here are just
a few! Many molecular
structures have the same shape of familiar space figures. Take another look at
the fullerene molecule shown below. The molecule has the same shape as an icosohedron.
Notice that the faces are composed of hexagons and pentagons. 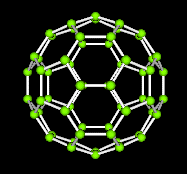
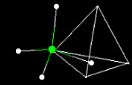
Below is shown the molecule
Methane. Try
this. Notice
when you rotate the methane molecule it is off-center. Click on the tools pulldown
and select 'pickcenter'. Click the center carbon atom in the methane molecule.
Now rotate the methane molecule. It should rotate around the center carbon atom.
What polyhedral
stucture would you think methane best represents? Click on the tetrahedral box.
Now rotate the object to show how the molecule fits snugly within the tetrahedron.
|
| | To
rotate object hold left mouse button down and slide cursor arrow over object |
| Below
is shown the molecule Benzene. Click off the hydrogens. What geometric pattern
do you see?
|
| | To
rotate object hold left mouse button down and slide cursor arrow over object |
| Do
you know what the interior angle measurements are for a hexagon? You can measure
the angles of the hexagon using the above software. First click the hydrogens
back on. Click on tools --> measures. Then click with the left mouse button
on three consecutive vertex points. The lower left hand corner will give the angle
measurement of 120 degrees along with several other measures. What is the sum
of the interior angles equal to? At the end of this textbook are several tutorials
which explain in greater detail how to use this software package and what all
those numbers at the lower left mean. Many
geometric figures display symmetry. 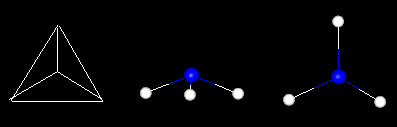 Image 5K Image 5K
The
equilateral triangle on the left displays rotational symmetry. Rotating the figure
about about a point formed by the intersection of the three angle bisectors (the
incenter of the triangle) will give the same appearance. The ammonia molecule
on the right also shows symmetry. It has a 3-fold axis of symmetry. Rotating the
molecule 120 degrees about its axis will give the same appearance.
Go to part II_Mathematical
Relationships |