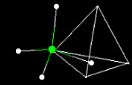
| What
will happen when these two water molecules approach each other? See
the Water Dimer Page for a Flash Animation. See
the Water Dimer Page for a Flash Animation.
What did you observe?
What are some properties of a single water molecule?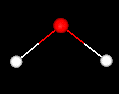
Each color
represents a different atom type. What type of atom is red? white? The
following images show a single water molecule after it has been rotated 90, 180,
270 and 360 degrees. 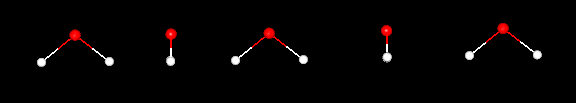
Does
water show any symmetry? Water
is formed when two atoms of hydrogen combine with one atom of oxygen. The single
electrons in the hydrogen atoms are drawn to oxygen which only has 6 electrons
in its valence shell. How will this affect the charge on the water molecule?
In
physics you learned that like charges repel and unlike charges attract. The above
animation shows what happens when two water molecules approach. What would expect
might happen if we surround a single molecule of water with four other molecules
of water? Take a look at this image frozen in time. 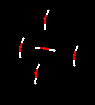 click
on image for pattern click
on image for pattern
Do
you recognize any familiar geometric pattern? What distances might each water
molecule be from each other? Can
you predict the pattern for 8 water molecules?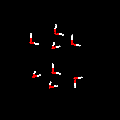 See image
See image The
above simulations were all performed at very low temperatures (below the freezing
point of water. At higher temperatures molecules are in rapid motion. What would
you expect the motion to look like at a higher temperature, say at room temperature?
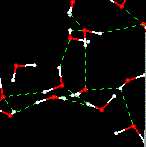 Movie Movie
|