| At
the molecular level symmetric patterns constantly show up.Even a single water
molecule shows symmetry.
Kinemage 1, which will appear on screen when lesson 4 is clicked on, shows a single
water molecule. **What
types of atoms is water composed of?____
**What is the chemical formula?_________ Click
on the ends to demonstrate the location of the hydrogens. The middle will show
the oxygen. **Measure
the distances from oxygen to either hydrogen. What value do you get?____________ **Is
the distance from either hydrogen to the oxygen the same?__
**What type of
symmetry does this represent?_______ It
is important to note that the units for the distance measurements are all in "Angstroms".
An Angstrom is 1/10,000,000,000 of a meter. Angstroms are always used when we
measuring at the atomic level. Can you give a reason why? Find
the angle represented for the water molecule on the screen.
Is it acute or
obtuse?_____ What is the measure?_______
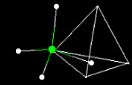
Click off the water molecule, and click on the methane molecule. A methane molecule
will appear. We have seen this shape before. **What
geometric shape do you think the methane will fit into?___________ Why?___________________ Click
on the tetrahedron box at the right side to demonstrate this. Leaving the tetrahedron
on. To find out what methane is composed clicking on each atom. Remember you must
click at the ends of the
lines. **Methane
is composed of _____________.
**The chemical formula for methane would be
_________. **Measure
the distance from carbon to each hydrogen?_________
**Measure the angles that
form the tetrahedron?___
_________________________________________________________________________________________ |