The Carbon Atom
Physical Properties of the Carbon Atom
Atomic
Number 6
Atomic Mass Average: 12.011
Melting Point: 3823 K (3550°C
or 6422°F)
Boiling Point: 4098 K (3825°C or 6917°F)
Density:
2.267g/cu.cm.
Velocity of sound [/m s-1]: 18350
Hardness Scale Mohs:
0.5
Stable Isomers (2)
Atomic Structure
The
Carbon atom has six electrons, 4 of the electrons are in its valence shell (outershell).
The circles in the diagram show energy levels - representing increasing distances
from the nucleus.
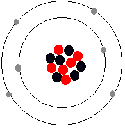 |
This diagram is, however, a simplification and can be misleading. It gives the impression that the electrons are circling the nucleus in orbits like planets around the sun. Actually it is not possible to know exactly where the electrons are located (see below) |
| A better way to look at the carbon atom is by using an energy level graph shown at the right. Here we see carbon has six electrons represented by arrows (the direction of the arrow represents the electron spin) Two electrons are found in the 1s orbital close to the nucleus. The next two will go into the 2s orbital. The remaining ones will be in two separate 2p orbitals. This is because the p orbitals have the same energy and the electrons would rather be in separate orbitals. | 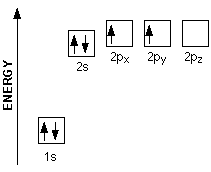 |
The actual location of electrons in a carbon atom cannot be determined with certainty and the electrons appear to be 'smeared' into orbitals as shown below. These images were created using the java applet --Atomic and Molecular Orbitals from MIT. This java applet and other Molecular Orbitals applets can be found at the Chemistry Java Page.
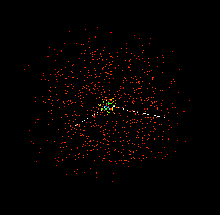 |
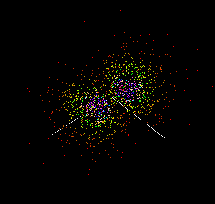 |
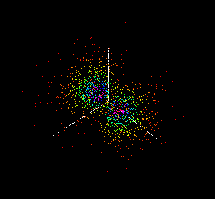 |
2s |
2px |
2py |
Isotopes
Isotopes are atoms which have the same atomic number but different mass numbers. They have the same number of protons but different numbers of neutrons.The number of neutrons in an atom can vary within small limits. For example, there are three kinds of carbon atom 12C, 13C and 14C. They all have the same number of protons, but the number of neutrons varies.
| protons | neutrons | mass number | |
| carbon 12 | 6 | 6 | 12 |
| carbon 13 | 6 | 7 | 13 |
| carbon 14 | 6 | 8 | 14 |
These different types of carbon atoms are called isotopes. The fact that they have varying numbers of neutrons makes no difference to the chemical reactions of the carbon atom.
Uses of Carbon
Graphite combined with clays form the 'lead'
used in pencils.
Diamond is used for decorative purposes, and also as drill
bits.
Carbon added to iron makes steel.
Carbon is used for control rods
in nuclear reactors.
Graphite carbon in a powdered, caked form is used as
charcoal for cooking, artwork and other uses.
Charcoal pills are used in
medicine in pill or powder form to adsorb toxins or poisons from the digestive
system.
Carbon Atoms and Molecules
- CARBON MODULE HOME
- THE CARBON ATOM
- CABON MOLECULE
- CARBON THE ELEMENT
- SIMPLE CARBON COMPOUNDS
- CARBON CHEMISTRY JMOL
- CARBON CHEMISTRY JSMOL
- GRAPHENE MOLECULE
- HOW BIG IS A BUCKEYBALL
- FULLERENE MOLECULE
- GRAPHITE AND DIAMOND
- GRAPHITE AND CARBYNE
- CARBON MONOXIDE
- GEOMETRY OF METHANE
- BENZENE MOLECULE
- CARBON FUELS
