SIMPLE CARBON COMPOUNDS
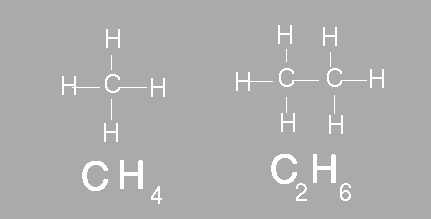
The simplest organic compounds contain molecules composed of carbon and hydrogen. The compound methane contains one carbon bonded to four hydrogens. Ethane is another example of a simple hydrocarbon. Ethane contains two carbon atoms and six hydrogen atoms. In chemistry we use a molecular formula to show how many atoms of each element are present in a molecule. A molecular formula however does not show the structure of the molecule. Scientists often use structural formulas to show the number and arrangement of atoms in a compounds. Below the molecular formula for methane and ethane are shown. Above the molecular formula are their respective structural formula.
Although structural formulas can be very helpful they do not give a complete picture of a molecule. Structural formulas do not tell us anything about the distances between bonds, the angles formed by these bonds, or the size and shape of the molecule. Scientists use three different representation to show what molecules look like.
THE WIRE FRAME MODEL
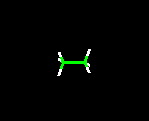 This model clearly shows the type of atoms in the molecule, the distances between
bonds, and angles associated with the atoms. Because the lines drawn are very
thin, molecules can very easily be manipulated when viewed on a computer screen.
This model clearly shows the type of atoms in the molecule, the distances between
bonds, and angles associated with the atoms. Because the lines drawn are very
thin, molecules can very easily be manipulated when viewed on a computer screen.
THE BALL-AND-STICK MODEL
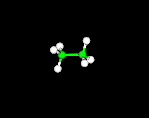 Atoms are represented by balls and bonds are represented as sticks.
Atoms are represented by balls and bonds are represented as sticks.
THE SPACE FILLED MODEL
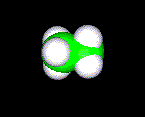 This model shows the space that the molecule will take up. Because of all
the points required to draw this molecule on a computer screen you should expect
these molecules to be very difficult to manipulate.
This model shows the space that the molecule will take up. Because of all
the points required to draw this molecule on a computer screen you should expect
these molecules to be very difficult to manipulate.
Other Carbon Compounds
| methane | ethane | propane | ||||||
|---|---|---|---|---|---|---|---|---|
| Gif | PDB | Gif | PDB | Gif | PDB | |||
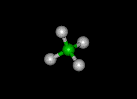 |
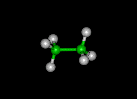 |
 |
||||||
| butane | pentane | hexane | ||||||
|---|---|---|---|---|---|---|---|---|
| Gif | PDB | Gif | PDB | Gif | PDB | |||
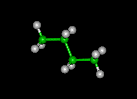 |
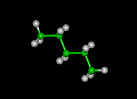 |
 |
||||||
| butadiene | benzene | cyclohexane | ||||||
|---|---|---|---|---|---|---|---|---|
| Gif | PDB | Gif | PDB | Gif | PDB | |||
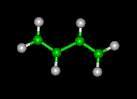 |
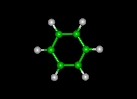 |
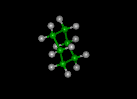 |
||||||
| ethylene | ethanol | dinitro | ||||||
|---|---|---|---|---|---|---|---|---|
| Gif | PDB | Gif | PDB | Gif | PDB | |||
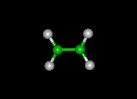 |
 |
 |
||||||
For a more complete list of carbon compounds see the MathMol Library of Molecules.
Carbon Atoms and Molecules
- CARBON MODULE HOME
- THE CARBON ATOM
- CABON MOLECULE
- CARBON THE ELEMENT
- SIMPLE CARBON COMPOUNDS
- CARBON CHEMISTRY JMOL
- CARBON CHEMISTRY JSMOL
- GRAPHENE MOLECULE
- HOW BIG IS A BUCKEYBALL
- FULLERENE MOLECULE
- GRAPHITE AND DIAMOND
- GRAPHITE AND CARBYNE
- CARBON MONOXIDE
- GEOMETRY OF METHANE
- BENZENE MOLECULE
- CARBON FUELS
